

The major research themes in our lab include near-infrared (NIR) emitting lanthanide materials for medical and optical applications, photoredox catalysts for efficient chemical conversion, and broadband light-harvesting materials for cost-effective solar energy unitilization.
Certain lanthanide ions, such as ytterbium(III), which emits at 980 nm, are promising candidates for the sensitive detection of tumor markers. Over the past two decades, numerous near-infrared (NIR)–emitting lanthanide complexes incorporating various sensitizers have been reported. However, their practical applications remain limited by low emission efficiencies and the reliance on UV or near-UV excitation sources. The goal of our research is to employ a molecular engineering strategy to develop novel lanthanide complexes that can be efficiently sensitized under red-light excitation. Specifically, we use porphyrin and BODIPY dyes as modular building blocks to construct stable, functionalized lanthanide complexes with enhanced photophysical properties
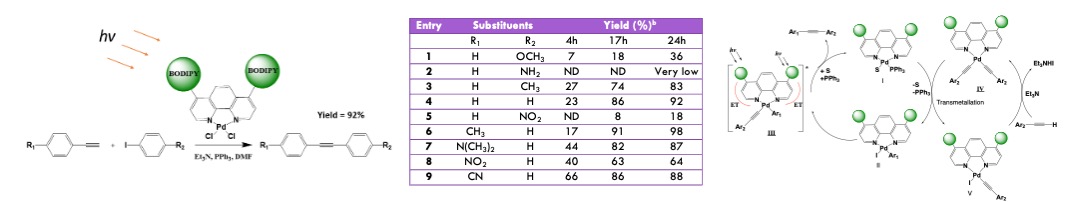
Photoredox catalysts are typically based on ruthenium or iridium complexes. When combined with conventional transition-metal catalysts such as palladium(II), nickel(II), or copper(II), these systems can promote a wide range of transformations, including C–C cross-coupling, C–N cross-coupling, and C–H arylation reactions. Depending on the specific photocatalyst and metal catalyst employed, these reactions may proceed via single-electron transfer (SET), energy transfer (EnT), or radical-mediated mechanisms. Our research focuses on developing “two-in-one” catalytic systems in which organic chromophores are directly integrated into a conventional metal catalyst. This design enables intramolecular SET or EnT processes, as illustrated in the following figure, and eliminates the need for separate photocatalyst and transition-metal catalyst components. We have demonstrated that a BODIPY-functionalized palladium(II) complex efficiently catalyzes Sonogashira C–C cross-coupling reactions under light irradiation.
Sunlight is abundant and renewable. It can be converted into other forms of stored energy, such as electricity, or used to drive chemical transformations, including the conversion of carbon dioxide into value-added products and the splitting of water into hydrogen and oxygen. Two major challenges in these processes are the narrow solar spectral coverage of light absorbers and the weak adsorption of dyes on electrode surfaces. For example, dye molecules must remain strongly bound to the surface of Pt-decorated TiO₂ nanoparticles during solar water splitting. We found that 8-hydroxyquinoline is an excellent anchoring group for dye-sensitized solar cells, as shown in the following graph. The goal of this research is to construct stable and efficient electrodes using well-designed broadband light absorbers for solar water splitting.

If you are interested in pursuing the aforementioned research in our group and would like to contribute to this field, please feel free to contact Dr. He by email for more information. We welcome applications from motivated undergraduate and graduate researchers.
