Origin of the Earth's
Atmosphere
Introduction
Early Earth would have been very different and inhospitable compared to
the Earth today.
-
Hot
-
Why? - Primordial heat, collisions and compression during accretion,
decay of short-lived radioactive elements
-
Consequences - Constant volcanism, surface temperature too high
for liquid water or life as we know it, molten surface or thin, unstable
basaltic crust.
-
Atmosphere - early atmosphere probably completely different in composition
(H2, He)
-
Cooling
-
Primordial heat dissipated to space
-
Condensation of water (rain), accumulation of surface water.
-
Accumulation of new atmosphere due to volcanic out gassing
-
Conditions appropriate for evolution of life
Evolution of the Atmosphere
Atmosphere - Envelope of gases that surrounds the Earth. Used by
life as a reservoir of chemical compounds used in living systems. Atmosphere
has no outer boundary, just fades into space. Dense part of atmosphere
(97% of mass) lies within 30 km of the Earth (so about same thickness as
continental crust).
-
Chemical Composition Today - Nitrogen (N2)- 78%, Oxygen (O2)-
21%, Carbon Dioxide (CO2) - 0.03 %, plus other miscellaneous
gases (H2O for one).
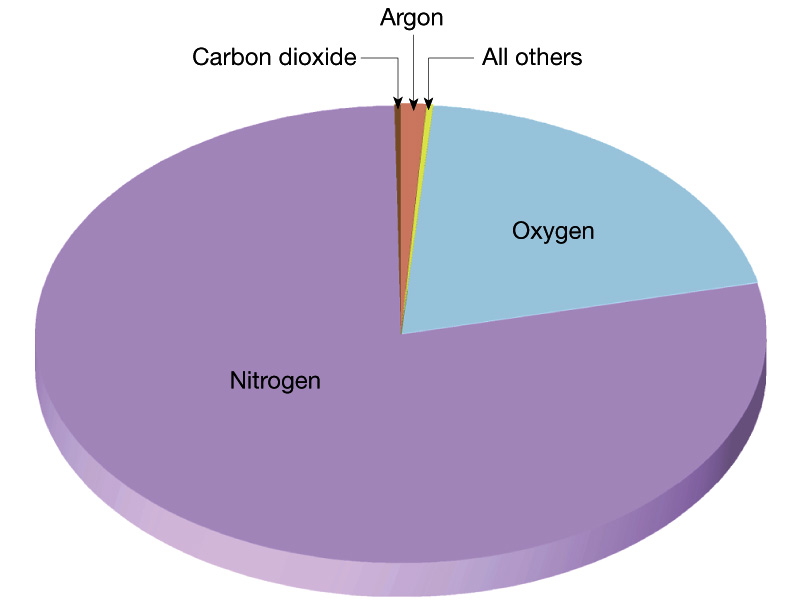
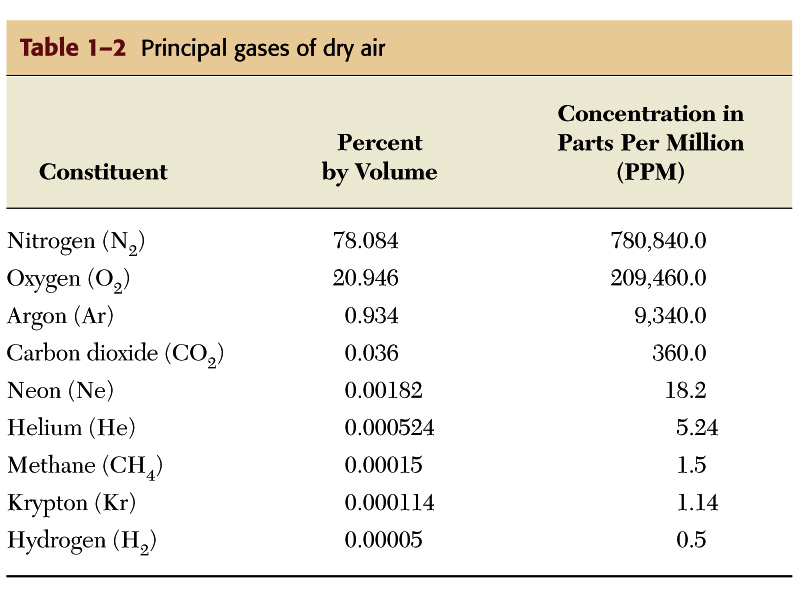
(figure and table from Lutgens and Tarbuck, The Atmosphere,
8th edition)
First Atmosphere
-
Composition - Probably H2, He
-
These gases are relatively rare on Earth compared to other places in the
universe and were probably lost to space early in Earth's history because
-
Earth's gravity is not strong enough to hold lighter gases
-
Earth still did not have a differentiated core (solid inner/liquid outer
core) which creates Earth's magnetic field (magnetosphere = Van Allen Belt)
which deflects solar winds.
-
Once the core differentiated the heavier gases could be retained
Second Atmosphere
Produced by volcanic out gassing.
-
Gases produced were probably similar to those created by modern volcanoes
(H2O, CO2, SO2, CO, S2, Cl2,
N2, H2) and NH3 (ammonia) and CH4
(methane)
-
No free O2 at this time (not found in volcanic gases).
-
Ocean Formation - As the Earth cooled, H2O produced by
out gassing could exist as liquid in the Early Archean, allowing oceans
to form.
-
Evidence - pillow basalts, deep marine seds in greenstone belts.
Addition of O2 to the Atmosphere
Today, the atmosphere is ~21% free oxygen. How did oxygen reach these levels
in the atmosphere? Revisit the oxygen cycle:
-
Oxygen Production
-
Photochemical dissociation - breakup of water molecules by
ultraviolet
-
Produced O2 levels approx. 1-2% current levels
-
At these levels O3 (Ozone) can form to shield Earth surface
from UV
-
Photosynthesis - CO2 + H2O + sunlight = organic
compounds + O2 - produced by cyanobacteria, and eventually higher
plants - supplied the rest of O2 to atmosphere. Thus plant populations
-
Oxygen Consumers
-
Chemical Weathering - through oxidation of surface materials (early
consumer)
-
Animal Respiration (much later)
-
Burning of Fossil Fuels (much, much later)
Throughout the Archean there was little to no free oxygen in the atmosphere
(<1% of presence levels). What little was produced by cyanobacteria,
was probably consumed by the weathering process. Once rocks at the surface
were sufficiently oxidized, more oxygen could remain free in the atmosphere.
During the Proterozoic the amount of free O2 in the
atmosphere rose from 1 - 10 %. Most of this was released by cyanobacteria,
which increase in abundance in the fossil record 2.3 Ga. Present levels
of O2 were probably not achieved until ~400 Ma.
Evidence from the Rock Record
-
Iron (Fe) i s extremely reactive with oxygen. If we look at the oxidation
state of Fe in the rock record, we can infer a great deal about atmospheric
evolution.
-
Archean - Find occurrence of minerals that only form in non-oxidizing environments
in Archean sediments: Pyrite (Fools gold; FeS2), Uraninite (UO2).
These minerals are easily dissolved out of rocks under present atmospheric
conditions.
-
Banded Iron Formation (BIF) - Deep water deposits in which
layers of iron-rich minerals alternate with iron-poor layers, primarily
chert. Iron minerals include iron oxide, iron carbonate, iron silicate,
iron sulfide. BIF's are a major source of iron ore, b/c they contain magnetite
(Fe3O4) which has a higher iron-to-oxygen ratio than
hematite. These are common in rocks 2.0 - 2.8 B.y. old, but do not form
today.
-
Red beds (continental siliciclastic deposits) are never found
in rocks older than 2.3 B. y., but are common during Phanerozoic time.
Red beds are red because of the highly oxidized mineral hematite (Fe2O3),
that probably forms secondarily by oxidation of other Fe minerals that
have accumulated in the sediment.
Conclusion - amount of O2 in the atmosphere has increased with
time.
Biological Evidence
-
Chemical building blocks of life could not have formed in the presence
of atmospheric oxygen. Chemical reactions that yield amino acids are inhibited
by presence of very small amounts of oxygen.
-
Oxygen prevents growth of the most primitive living bacteria such as photosynthetic
bacteria, methane-producing bacteria and bacteria that derive energy from
fermentation. Conclustion - Since today's most primitive life forms are
anaerobic, the first forms of cellular life probably had similar metabolisms.
-
Today these anaerobic life forms are restricted to anoxic (low oxygen)
habitats such as swamps, ponds, and lagoons.
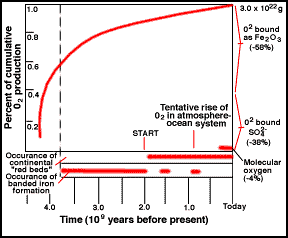
Atmospheric oxygen built up in the early history of the Earth as the
waste product of photosynthetic organisms and by burial of organic matter
away from surficial decay. This history is documented by the geologic preservation
of oxygen-sensitive minerals,
deposition banded iron formations, and development of continental "red
beds" or BIFs. Figure from the University of Michigan's Introduction to
Global Change web site.
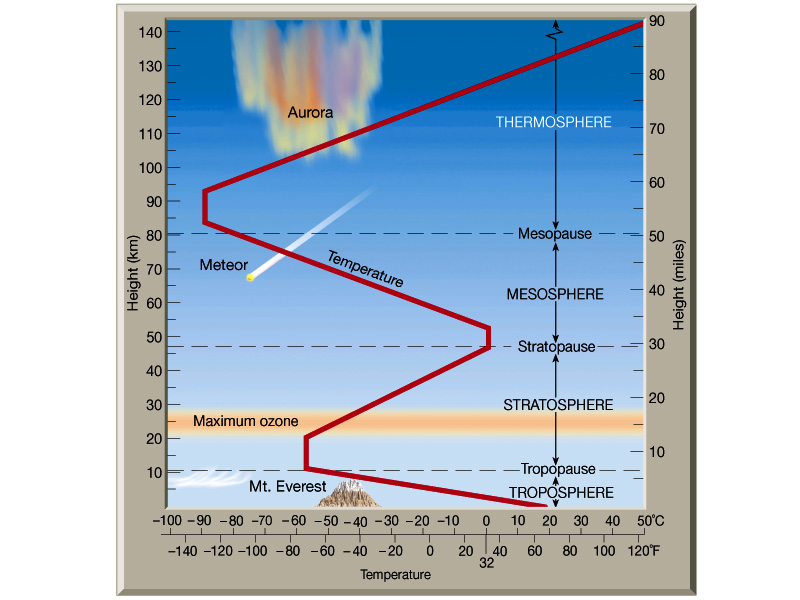
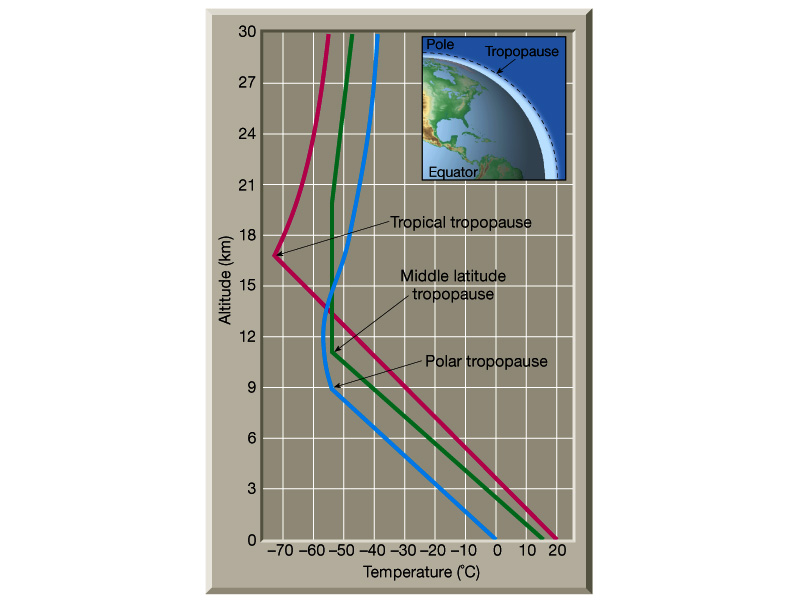
Atmospheric Structure
Not only does the atmosphere have a relatively stable composition,
but it also has a structure to it. Below are two figures from Lutgens
and Tarbuck (The Atmosphere, 8th edition) showing vertical profiles
of the atmosphere. Note that the first major break, or boundary in
the atmosphere as we ascend is the tropopause.