PHY1161
Exam #4
April 30, 2003
Statistics:
High = 91
Mean = 72
Low = 31
| Return to Calendar Page |
Possibly useful information:
c = 3.00 x 108 m/s h = 6.626 x 10–34 J s = 4.14 x 10 - 15 eV s
Be sure to bring your own equations sheet for the FINAL exam!
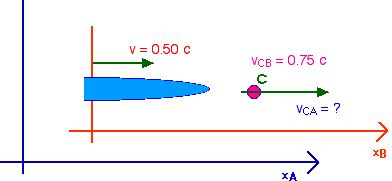
1. [20 pts] A spaceship travels away from
Earth at a velocity of v = 0.5 c. A projectile is shot forward from the spaceship
at vCB = 0.75 c relative
to the spaceship. What is the velocity of this projectile relative
to Earth. vCA ?

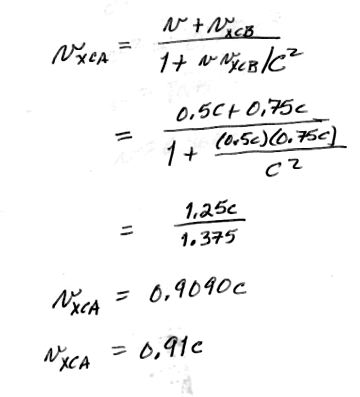
2. [15 pts] How fast must a meter stick be moving to appear only 0.5 m long to an Earth-based observer?
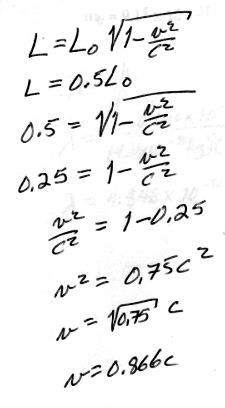
3. [10 pts] What is the deBroglie wavelength of an electron moving at 1.5 x 108 m/s (v = 0.5 c)?
me = 9.11 x 10 - 31 kg
4. [10 pts]
What is the energy of the photons in the visible region of the spectrum?
(Use ![]() = 550 nm as
an average value).
= 550 nm as
an average value).
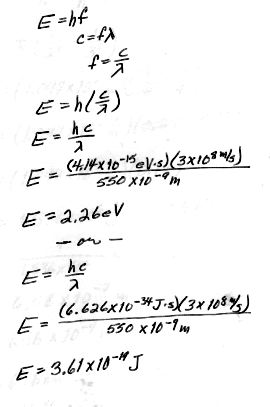
5. [10 pts] Calculate the first wavelength of the Balmer series for hydrogen and tell what color it is.
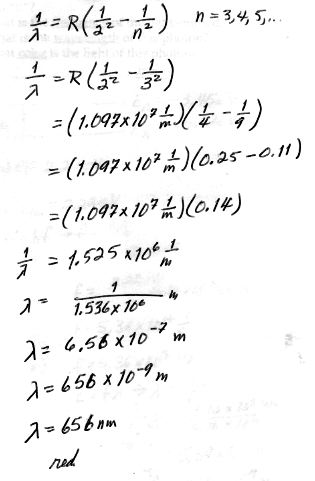
6. [20 pts] In state “A” the energy of an atom is – 3.45 eV and in state “B” its energy is – 5.67 eV.
a) What is the energy of the photon emitted in the transition from “A” to “B”?
b) What is the frequency of the light emitted?
c) What is the wavelength of this photon?
d) What color is the light of this photon?

7. [0 pts] My wife calls me a “blithering optimist”. Earlier last week I had expected/hoped to say a few words about Nuclear Physics. I was unable to talk that fast. Here is the question I would have included.
Carbon-14 {14C) is radioactive by ordinary beta-decay. What is the daughter nuclide produced when Carbon-14 {14C) decays?
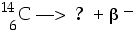
We need to balance the charges and the nucleon masses. It will probably help to write those numbers for the beta (b) particle directly as

Now it should be easy to see that the daughter nucleus must have a mass number A of 14 and must have Z = 7 protons in the nucleus. From the Periodic Table at the front of the room you can find that Z = 7 means the daughter nuclide is Nitrogen (N). Therefore, we can write this as
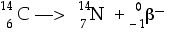
and if you wanted to re-write it with the beta(b) stripped of its numbers, you would then have

In any case the daughter nuclide is Nitrogen-14.
CQ. [15 pts] Concept Questions:
a) E = m c2 is a very famous equation -- perhaps even as
well known as Newton’s F = m a. What does this equation “ E = m c2
” mean?
Energy and mass are the same thing. Mass can be converted into energy -- this
is the basis of nuclear power.
b) What is the photoelectric effect?
When light shines on a metal, electrons may be ejected from the metal.
c) What is meant by “the complementarity principle” or “wave-particle
duality”?
For both light and electrons (and other small “particles”) explaining
them in terms of waves or in terms of particles had seemed contradictory.
After many years of work, trying to reconcile these two different descriptions
or theories, Niels Bohr concluded that the two modles are not contradictory.
Rather, he said, they are complementary -- the two theories complement
each other. Both theories are necessary to describe light or electrons.
d) What element is the most maleable? That is, what element can be
processed into the thinnest sheet?
Gold
That is why Rutherford use a very thin gold foil in his scattering experiments.
Since gold could be made so thin, he only had to consider a single scattering
event from a single gold atom.
e) What happens when an electron moves from one “stationary orbit”
to another?
The energy lost by the electron as it moves to a lower-energy orbit is given
up as the energy of an out-going photon of light. For an electron to move to
a higher-energy orbit, light of just the right energy (or frequency or wavelength)
must come in and be absorbed by the atom to provide that energy.