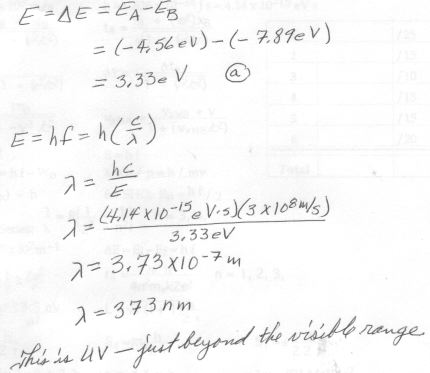PHY1161
Exam #4
December 11, 2002
1. [25 pts] (27.2) A flashbulb goes off at the origins as the origins of our usual reference frames A and B coincide. Frames A and B have a relative speed of 0.6 c along their x-axes. A wavefront travels along the x-axis as observed in B. What time is recorded in B as this wavefront reaches xB = 1,000 m? Use these values of xB and tB to calculate the coordinates xA and tA that describe this event in A. Then use these values to calculate the speed of light in reference frame A ( vlight = xA/tA).
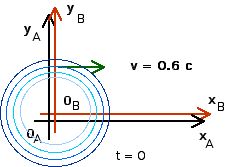
To find time tB we just use the definition of velocity
v = c = xB/tB
tB = xB/c
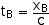
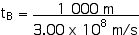
tB = 3.333 x 10–6 s
Of course, we also have
xB = 1000 m
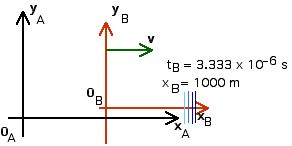
Now we can apply the Lorentz Transformations to find xA and tA,
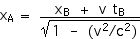
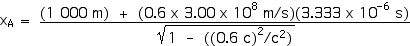
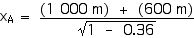
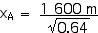
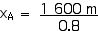
xA = 2 000 m
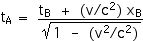

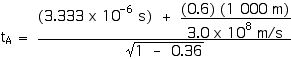
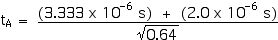
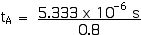
tA = 6.66 x 10–6 s
Now we can find the velocity in the A-frame and determine if the Lorentz Transformations work. Is it, indeed, true that the velocity of light will be the same in both frames? Let’s see, . . .
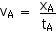
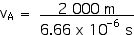
vA = 3.00 x 108 m/s
And, indeed, this is the speed of light!
==========================================================
2. [15 pts] (27.49) A spaceship travels away from Earth at a velocity of 2 x 108 m/s. A projectile is shot forward from the spaceship at 2.5 x 108 m/s relative to the spaceship. What is the velocity of the projectile relative to Earth?
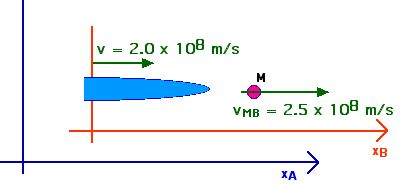
This provides a more direct application of our velocity transformation equation,
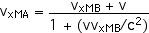
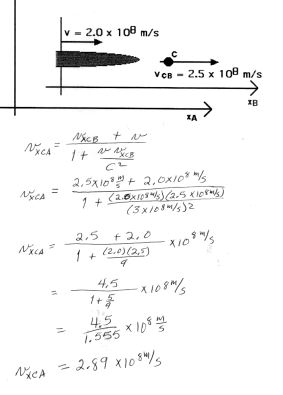
==========================================================
3. [10 pts]

==========================================================
4. [15 pts]

==========================================================
5. [15 pts] (29.1) Calculate the second wavelength of the Balmer series for hydrogen and tell what color it is.
Balmer Series for Hydrogen
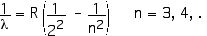
R = 1.097 x 107 m –1, Rydberg constant
For n = 4 we have
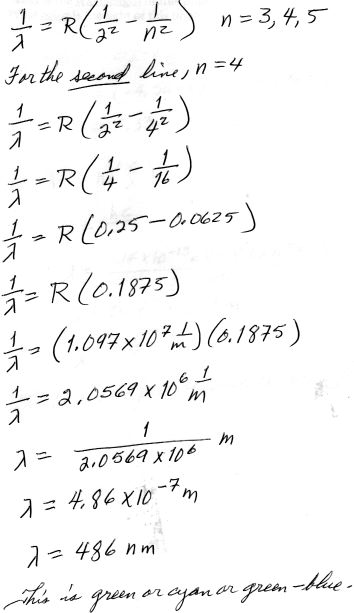
==========================================================
6. [20 pts] (29.16) In state “A” the energy of an atom is – 4.56 eV and in state “B” its energy is – 7.89 eV.
a) What is the energy of the photon emitted in the transition from “A” to “B”?
b) What is the wavelength of this photon?
c) What color is the light of this photon?