
Ideal Gases
![]() An
ideal gas obeys PV/T = constant or PV/T =
PoVo / To exactly.
An
ideal gas obeys PV/T = constant or PV/T =
PoVo / To exactly.
![]() P,
V, and T are known as "state variables" for specifying two of them
fully determines the "state" or condition of an ideal
gas.
P,
V, and T are known as "state variables" for specifying two of them
fully determines the "state" or condition of an ideal
gas.
![]() The
ideal gas law can be written as PV = nRT where n specifies how
much gas is being considered and R is a universal
constant.
The
ideal gas law can be written as PV = nRT where n specifies how
much gas is being considered and R is a universal
constant.
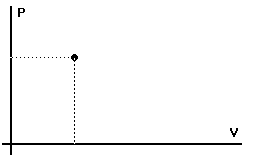
"State variables"
![]()
![]()
![]()
This is known as the ideal gas law.
k = Boltzman's constant
k = 1.38 x 10-23 J/K
n = number of moles
A mole of gas molecules is Avagadro's number NA of molecules,
[ N = n NA ]
R = universal gas constant
R = 8.314 J/mole-K
For constant pressure, an increase in
temperature brings about an increase in volume, according to
![]()
|
|
|
|
|
|
|
|
||
|
|
|||
