

| ToC, Chapter 17 | Course Calendar |
D17.1 A goose-down sleeping bag has a surface area of 2.25 m2 and is filled with a layer of down 5 cm thick. What is the heat-transfer rate (Hcd) through it from a person with skin temperature of 35°C to the outside air at -5°C. How does this rate compare with the body’s minimal metabolism rate, about 100 W?
The thermal conductivity of goose down isK = 0.023 W/m C° We can now apply the conduction heat-transfer equation, Eq 13.1,
Hcd = [K A (T2 - T1)] / L From the problem, we know the other pieces of this equation,
DT = T2 - T1 = 35°C - (- 5°C) = 40 C°
A = 2.25 m2
L = 5 cm = 0.05 m
Hcd = [K A (T2 - T1)] / L
Hcd = [(0.023 W/m C°)(2.25 m 2)(40 C°)]/0.05 m =
Hcd = 41 WThis is about half of the body’s normal metabolism so this sleeping bag should keep our camper a “happy camper” (or, at least, a warm camper).
(Hmmm, . . . go ahead and use e = 1.00, even in this case).
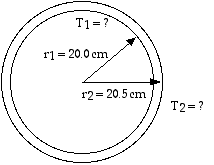
To find the temperature, we can use the Radiation equation, Eq 13.4,Remit = e A T 4
We will use e
1.0, even for this. s is the Stefan-Boltzman constant,
= 5.67 x 10 - 8 W/m2 K4
The surface area of a sphere is
A = (4) r 2
For the inner surface,
Ainner = (4) (0.200 m)2 = 0.50265 m2
For the outer surface,
Aout = (4) (0.205 m)2 = 0.5281 m2
The temperatures T must be absolute temperatures, measured on the Kelvin scale.
Now we are ready to solve for the temperatursT 4 = Remit / e A
Tinner4 = 100 W/[(1.0)(5.67 x 10 - 8 W/m2 K4)(0.50265 m2)] = 3.509 x 10 9 K4
Tinner = 243 K
Touter4 = 100 W/[(1.0)(5.67 x 10 - 8 W/m2 K4)(0.5281 m2)] = 3.3396 x 10 9 K4
Touter = 240 K
Our use of e = 1.0 , for the emissivity, makes our actual numbers rather “iffy”. But the point of this problem was to show that the inner surface will be at a higher temperature than the outer surface. That is certainly true for our calculation. Changing the value of the emissivity e may change our actual numbers but we will still find Tinner > Touter.
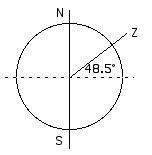
N and S mark the poles, the axis of rotation. Z is the zenith, the point directly overhead, 48.5° from the equator (48.5° north latitude).
Summer:
is the angle from directly overhead (the zenith) to the Sun. Therefore, the angle between the horizon and the Sun will be 90° -
= 90° - 25° = 65°. The Sun will be 65° above the horizon at the summer solstice.
Winter:
is again the angle between directly overhead (the zenith) and the Sun. Therefore, the Sun is 90° - 72° = 18° above the horizon at the winter solstice.
The first part of this questions requires exactly the same reasoning, using exactly the same kind of diagrams, as question 13.21.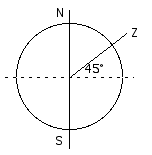
N and S mark the poles, the axis of rot ation. Z is the zenith, the point directly overhead, 45° from the equator (45° north latitude).
Summer: 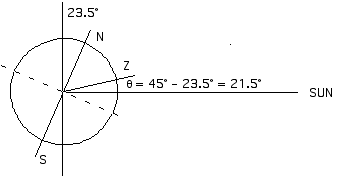
If the Sun is 21.5° from the zenith (the position directly overhead) then it must be 90° - 21.5° = 68.5° above the horizon.
Winter: 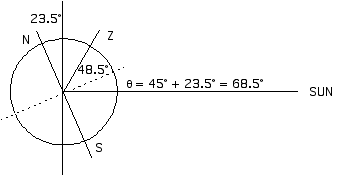
If the Sun is 68.5° from the zenith, then it must be 90° - 68.5° = 21.5° above the horizon.
Now we can look at the shadow cast by the eave on these two days:
Summer: 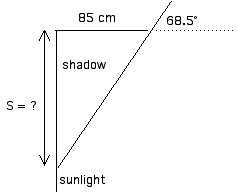
The length of the shadow, S in the diagram, is the opposite side of a 68.5° right triangle (85 cm is the adjacent side).
tan = opp/adj
tan 68.5° = S / 85 cm
S = (85 cm)(tan 68.5°)
S = (85 cm)(2.54) = 216 cm = 2.16 cmWinter:
The length of the shadow, S in the diagram, is the opposite side of a 21.5° right triangle (85 cm is the adjacent side).
tan = opp/adj
tan 21.5° = S / 85 cm
S = (85 cm)(tan 21.5°)
S = (85 cm)(0.393) = 33.5 cm
********************************************************
CHANGE NEEDED
********************************************************
D17.5 Calculate the R value of an insulated wall like that in Figure 13.30 if 2 x 6’s are used for wall studs instead of 2 x 4’s. The larger board allow 5.5 in of fiberglass insulation instead of 3.5 in.By what percentage does this increase in thickness change the R value?
How does the increased thickness change the heat loss calculated in Example 13.14?
The R-values for the components of the wall in Figure 13.30 are given in Table 13.4, on page 481. By increasing the fiberglass insulation from 3.5 inches to 5.5 inches, the R-value for this insulation increases from 10.90 to 18.80, according to Table 13.3, on page 480. This increases the total R-value from 14.33 to 22.23.
This is an increase of 7.9/14.33 = 0.55 = 55% for the R-value of the wall.
In terms of R-values, the conduction heat transfer rate is given by equation 13.9,Hcd = A (T2 - T1) / R = (1/R) [A (T2 - T1)] In Example 13.14, we found the conduction heat transfer rate to be
Hcd(un) = 3.8 kW for the uninsulated wall,
Hcd(3.5) = 1.2 kW for the insulated wall with 2x4’s and 3.5 in of insulation
Now, with 2x6’s and 5.5 in of insulation,Hcd(5.5) = (1/22.23)[(960)(60)] = 2591 Btu/h = 0.76 kW This is a reduction of heat flow by
H = 1.18 kW - 0.76 kW = 0.42 kW
H / H = 0.42 kW / 1.18 kW = 0.36 = 36%
D17.6 A roasted turkey cools from 85°C to 80°C in 10 min
when sitting in a 25°C room. How long does it require to cool from 85°C
to 55°C?
This requires an application of Newton’s Law of Cooling,T(t) = Tsur + T e- t/

T is the initial temperature difference of the turkey and its surroundings;
T = 85°C - 25°C = 60 C°
Knowing that it cools from 85°C to 80°C allows us to solve for the “time constant” t in this equation;
T(t) = Tsur + T e-t/
T(10 min) = 80°C = 25°C + (60 C°) e-( 10 min)/
55°C = (60 C°) e- (10 min)/
55°C/60°C = e- (10 min)/
55/60 = e- (10 min)/t
0.9167 = e- (10 min)/
e- (10 min)/= 0.9167
ln[e- (10 min)/] = ln[0.9167]
- (10 min)/= -0.0870
10 min/= 0.0870
= (10 min)/0.0870
= 115 min
Now we know the time constant t and we can use Newton’s Law of Cooling to go back and solve for t, the time, when T, the temperature, is 55°C.
T(t) = Tsur +
T e- t/
T(t) = 55°C = 25°C + (60 C°) e-t /(115 min)
55°C = 25°C + (60 C°) e- t/(115 min)
30°C = (60 C°) e- t/(115 min)
30°C/60°C = e- t/(115 min)
30/60 = e- t /(115 min)
0.50 = e- t/(115 min)
ln[0.50] = ln[e- t/(115 min)]
-0.693 = -t/(115 min)
0.693 = t/(115 min)
t = (0.693)(115 min)
t = 78 min
D17.7 If concrete roadway sections are poured butted against each other when the temperature is 15°C, what will the thermal stress be when the temperature reaches 40°C? Young’s modulus for concrete is about 20 x 109 Pa.
The thermal stress is given byF/A = Y
T
From the text, we have the coefficient of linear expansion for concrete,
= 12 x 10-6 (C°) - 1
F/A =Y
T
F/A = [12 x 10-6 (C° ) - 1 ] [20 x 109 Pa] [25 C°]
F/A = 6 x 106 Pa
D17.8 If 0.60 kg of boiling hot coffee is poured into a 0.250-kg steel camping cup initially at 20°C, what is the final temperature of the cup-coffee system?
Q1 + Q2 = 0
Q1 = Qcup = ccup mcupTcup = csteel msteel
Tsteel
From Table 12.2, p 438, we find that the specific heat of steel is csteel = 450 J/kg C°
Q1 = csteel msteel Tsteel = ( 450 J/kg C° ) (0.250 kg) (Tf - 20°C)
Q2 = Qcoffee = ccoffee mcoffeeTcoffee = cwater mwater
Twater
Q2 = cwater mwaterTwater = (4186 J/kg C°) (0.60 kg) (Tf - 100°C)
where we have taken the specific heat of coffee to be the same as the specific heat of water,
ccoffee = cwater = 4186 J / kg C°.
Now we can put these all together as
Q1 + Q2 = 0
( 450 J/kg C° ) (0.250 kg) (Tf - 20°C) + (4186 J/kg C°) (0.60 kg) (Tf - 100°C) = 0
112.5 (J/C°) Tf - 2,250 J + 2,512(J/C°) Tf - 251,200 J = 0
112.5 (J/C°) Tf + 2,512(J/C°) Tf = 2,250 J + 251,200 J
[ 112.5 + 2,512 ](J/C°) Tf = [ 2,250 + 251,200 ] J
2,624.5 Tf = 253,450 °C
Tf = 253,450 °C/2,624.5
Tf = 96.6°C
D17.9 A 0.050-kg ice cube, initially at - 5.0°C, is placed in 0.30 kg of water at 25°C. What is the final temperature of the water? Or, if melting is not complete, how much ice remains unmelted at thermal equilibrium? Assume no heat is lost to the environment.
First, assume all the ice melts, so Tf > 0°CQ1 + Q2 = 0
Q1 = heat lost by the water
Q1 = cwater mwaterTwater = (4186 J/kg C°)(0.30 kg)(Tf - 25°C)
Q1 = (4186 J/kg C°)(0.30 kg)(Tf - 25°C) = 1,256 (J/C°) Tf - 31,395 JQ2 = heat gained by ice
Q2 = heat gained in raising ice to 0°C +
+ heat gained in melting ice +
+ heat gained in raising the melted ice (now water) to temperature Tf
Q2 = cice miceT1 + Lf,ice mice + cwater mice
T2
Q2 = [2090 J/kg C°][0.05 kg][0°C - (- 5°C)] +
+ [3.33 x 105 J/kg][0.05 kg] +
+ [4186 J/kg C°][0.05 kg][Tf - 0°C]Be very careful with these temperature changes!
Q2 = 522.5 J + 16,650 J + 209.3 (J/C°) Tf - 0
Q2 = 17,172.5 J + 209.3 (J/C°) TfNow we are ready to go back to
Q1 + Q2 = 0
1,256 (J/C°) Tf - 31,395 J + 17,172.5 J + 209.3 (J/C°) Tf = 0
1,256 (J/C°) Tf + 209.3 (J/C°) Tf = 31,395 J - 17,172.5 J
1,465 (J/C°) Tf = 14,222 J
Tf = [14,222/1,465]°C
Tf = 9.7°CAnd this is a reasonable answer.
D17.10 A 0.15-kg ice cube initially at -15°C is placed in 0.30 kg of water at 25°C. What is the final temperature of the water? Or, if melting is not complete, how much ice remains unmelted at thermal equilibrium? Assume no heat is lost to the environment.
This should look very similar to the previous question or problem. Now we have a larger and colder ice cube.
First, assume all the ice melts, so Tf > 0°CQ1 + Q2 = 0
Q1 = heat lost by the water
Q1 = cwater mwaterTwater = (4186 J/kg C°)(0.30 kg)(Tf - 25°C)
Q1 = (4186 J/kg C°)(0.30 kg)(Tf - 25°C) = 1,256 (J/C°) Tf - 31,395 J
Q2 = heat gained by ice
Q2 = heat gained in raising ice to 0°C +
+ heat gained in melting ice +
+ heat gained in raising the melted ice (now water) to temperature Tf
Q2 = cice miceT1 + Lf,ice mice + cwater mice
T2
Q2 = [2090 J/kg C°][0.15 kg][0°C - (- 15°C)] +
+ [3.33 x 105 J/kg][0.15 kg] +
+ [4186 J/kg C°][0.15 kg][Tf - 0°C]Be very careful with these temperature changes!
Q2 = 4,702 J + 49,950 J + 628(J/C°)Tf - 0
Q2 = 54,652 J + 628(J/C°)Tf
Q1 + Q2 = 0
1,256 (J/C°) Tf - 31,395 J + 54,652 J + 628 (J/C°)Tf = 0
[1,256 + 628](J/C°)Tf + [- 31,395 + 54,652]J = 0
1,884](J/C°)Tf + 23,257J = 0
1,884](J/C°)Tf = - 23,257J
Tf = [- 23,257/1,884]°C
Tf = - 12.3°CAnd this is inconsistent with our assumption that Tf > 0. Therefore, we have to look at another possibility. This time, we shall assume Tf = 0°C and only mmelt of the ice melts. Our calculation for Q1 is almost the same as before but our calculation for Q2 is quite different.
Q1 = heat lost by the water
Q1 = cwater mwaterTwater = (4186 J/kg C°)(0.30 kg)(0°C - 25°C)
Q1 = (4186 J/kg C°)(0.30 kg)(- 25°C) = - 31,395 J
Q2 = heat gained by ice
Q2 = heat gained in raising ice to 0°C + heat gained in melting some ice
Q2 = cice miceT1 + Lf,ice mmelt
Q2 = [2090 J/kg C°][0.15 kg][0°C - (- 15°C)] + [3.33 x 105 J/kg][mmelt]
Q2 = 4,702 J + 3.33 x 105 (J/kg)mmelt
Q1 + Q2 = 0
- 31,395 J + 4,702 J + 3.33 x 105 (J/kg)mmelt = 0
- 26,693 J + 3.33 x 105 (J/kg)mmelt = 0
3.33 x 105 (J/kg)mmelt = 26,693 J
mmelt = [26,693/333,000] kg
mmelt = 0.080 kgSince we started with 0.150 kg, this is a reasonable answer. There will remain 0.070 kg of ice that has not melted.
D17.11 The inside diameter of a steel lid and the outside diameter of a glass peanut butter jar are both exactly 11.50 cm at room temperature, 21.0°C. If the lid is stuck and you run 80°C hot water over it until the lid and the jar top both come to 80°C, what will the new diameters be?
For the steel lid, the change in the diameter,d, is
d =
do
T = [12 x 10- 6(C°)-1] [11.50 cm] [59 C°] = 0.0081 cm
so the new diameter is
dnew = do + d = 11.5081 cm
For the glass jar, the change in the diameter, Dd, is
d =
do
T = [10 x 10- 6(C°)-1] [11.50 cm] [59 C°] = 0.0068 cm
so the new diameter is
dnew = do + d = 11.5068 cm
Now the difference in the two diameters is 0.0013 cm. That may be enough to break the lid loose and let us make our peanut butter sandwich!
D17.12 If concrete roadway sections are poured butted against each other when the temperature is 15°C, what will the thermal stress be when the temperature reaches 40°C? Young’s modulus for concrete is about 20 x 109 Pa.
The thermal stress is given byF/A = Y
T
From the text, we have the coefficient of linear expansion for concrete,
= 12 x 10-6 (C°) - 1
F/A =Y
T
F/A = [12 x 10-6 (C° ) - 1 ] [20 x 109 Pa] [25 C°]
F/A = 6 x 106 Pa
| Return to Calendar |
(c) 2005, Doug Davis; all rights reserved